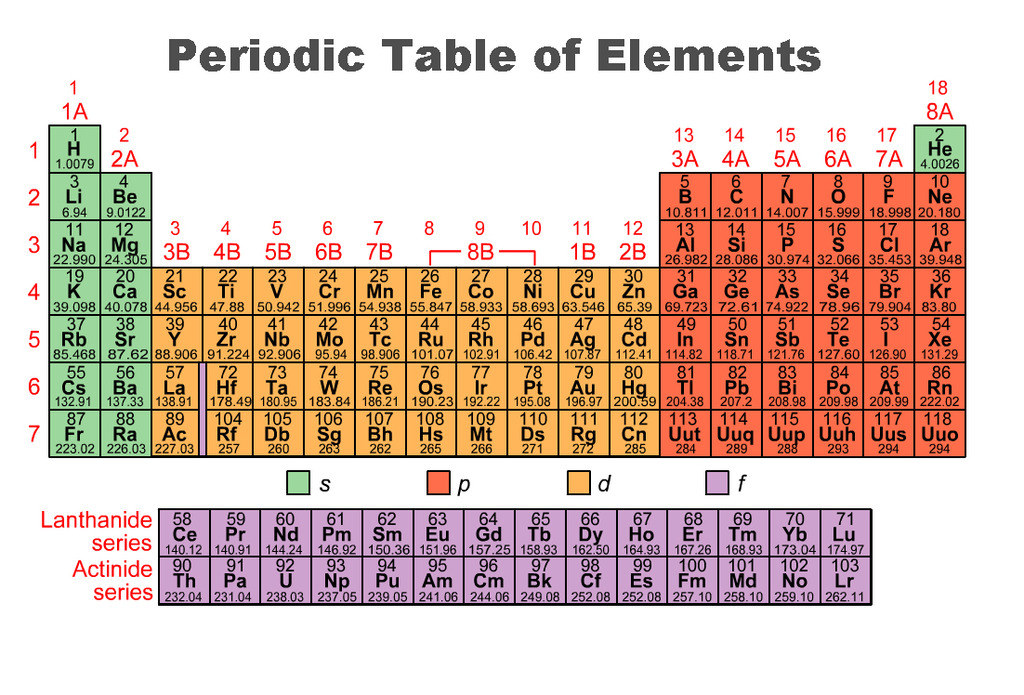
-Periods on a periodic table are the rows from left to right. They tell you the energy levels of the elements.
-Groups are up and down rows and they tell you the valence electrons.
-Valence electrons? You can find them on the top numbers above each row.
-Metals? They are found mostly in the middle and bottom section.
-Nonmetals? They are found on the right side of the table and hydrogen is also a nonmetal (1st element)
-Metalloids? are found as a stair-step like outline Including: B, Si, Ge, Sb, As, Te, At
-Atomic Numbers? They are the numbers on the top of all the elements, they represent the number of protons and electrons.
-Atomic mass? Are the bottom numbers on the elements (usually contain decimals).It tells us the mass of the element.
-How Reactive? You can tell how reactive an element is if it's in the first group rather than the last group.
-
-Groups are up and down rows and they tell you the valence electrons.
-Valence electrons? You can find them on the top numbers above each row.
-Metals? They are found mostly in the middle and bottom section.
-Nonmetals? They are found on the right side of the table and hydrogen is also a nonmetal (1st element)
-Metalloids? are found as a stair-step like outline Including: B, Si, Ge, Sb, As, Te, At
-Atomic Numbers? They are the numbers on the top of all the elements, they represent the number of protons and electrons.
-Atomic mass? Are the bottom numbers on the elements (usually contain decimals).It tells us the mass of the element.
-How Reactive? You can tell how reactive an element is if it's in the first group rather than the last group.
-